  
Therefore, after complete n half-lives, the amount of radioactive element reaming (1/2) n. Hence, after 8 hours it decomposes 75% and reaming 25% and the process continued. After four hours, it decomposes 50% and the remaining 50%. If the radioactivity of an element 100% and the half-life period of this element 4 hours. The half-life of polonium-213 isotope = 4.2 × 10 -6 sec and bismuth-209 isotope = 3 × 10 7 years. 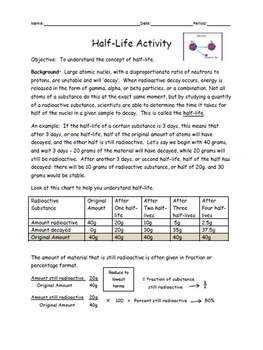
Hence the half-life is independent of the amount of the radio element present at a given time. This equation provides the relation between half-life and radioactive decay constants. The radioactive decay formula, ln(N 0/N) = kt but when t = t ½, N = N 0/2. 
Half-life is a characteristic property of radioactive elements. This period of decay is called the half-life of radioactive material or substances. What is half-life period?Īfter a certain period of time, the value of (N 0/N ) becomes one-half of the radioactive elements. The negative sign shows that N decreases with time. Therefore, the radioactive decay constant represents the fraction of radioactive atoms that disintegrates in unit time. The quantity dN/N represents the fraction of the total number of atoms that disintegrate in time dt. The radioactive decay formula is given below the picture,įrom the above formula, – (dN/N)/dt = k = rate constant. If N 0 number of the atoms present at the time t = 0 and N number of atoms present after the time t. Therefore, dN/dt = – kN, where N = number of the atoms of the disintegrating radioactive element, dt = time over which the decay is measured, and k = radioactive decay constant. The rate of radioactive decay formula depends only on the single power of the radioactivity. It is used mainly in radiocarbon dating and the determination of the age of matter or materials. Knowledge of half lives is part of how geologists date rocks with radioisotopic dating.Radioactive decay is one important natural phenomenon obeying the first-order chemical kinetics. It is important to note that regardless of the actual length of the half-life (whether it is millions of years or a few nanoseconds) the shape of the graph will be the same. The graph shown in Figure 1 is a visual representation of these equations above. N = N_o \left(\frac is the half life of the substance This is determined from properties such as the half life of the substance, and how much of the substance there was initially. There is an equation that is frequently used to determine how much of a certain radioactive substance remains after a given time has passed. Uranium-235 (another naturally occurring isotope of uranium) has a shorter half life than uranium-238, that's only ~700 million years. This means that it would take billions of years for uranium-238 to decay into a ratio of half uranium-238 and half thorium-234. Uranium-238 has a half life of 4.51 billion years. In contrast, some elements have extraordinarily long half lives and take billions of years to decay. For example, krypton-101 has a half life of about a ten millionth of a second. Some radioactive elements have half of their unstable nuclei decay in much less than one second. ĭifferent substances experience a loss of their radioactivity more quickly than others. Since half life is a measure of time, the half life is a value that determines how long this reduction to a more stable energy state will take. This results in an emission of some form of energy that travels along a 'ray', which is why we call it radiation. The stable nuclei in the sample are unchanging (and in a stable energetic state), but the unstable nuclei will undergo some sort of nuclear decay over time to become stable. In addition, there are also some nuclei within the substance that are already in their stable state but the proportion of stable to unstable nuclei in a sample can vary. These radioactive atoms release energy to become new, different types of atoms at some measurable rate known as radioactive decay.Īll radioactive materials have unstable nuclei within them these are nuclei that will decay. This also implies that one half life is the time that it takes for the activity of a source to fall to half its original value. Half life is the time that it takes for half of the original value of some amount of a radioactive element to decay. The time that it takes the mass or activity of the source (the number of decay events per second) to fall to the 50% mark is the half life. A chart showing the decay of a radioactive nucleus over time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
